Neuroimaging insights into brain mechanisms of early-onset restrictive eating disorders
Sample characteristics
Our analysis included 290 participants (124 EO-AN, 50 ARFID and 116 TD) (Table 1). As expected, EO-AN and ARFID had a significantly lower mean z-scored BMI compared with TD (BMI of −1.81 (95% confidence interval (CI) −4.1 to 0.61) for EO-AN, −2.20 (95% CI −4.2 to 0.38) for ARFID and 0.39 (95% CI −2.27 to 2.5) for TD, P = 2 × 10−16. rEO-EDs were slightly older than TD (EO-AN 11.5 ± 1.19 years, ARFID 11.2 ± 1.49 years and TD 10.2 ± 2.04 years, P = 9.8 × 10−8), with no age differences between EO-AN and ARFID (P = 0.6). As expected, there were differences in sex distribution (106 F/18 M for EO-AN, 38 F/12 M for ARFID and 59 F/57 M for TD, P = 2.2 × 10−8). Sensitivity analyses conducted on female individuals only showed no differences compared with the results obtained from the whole dataset (Supplementary Fig. 3).
Thinner cortex and smaller SVs in EO-AN > TD
Global effects
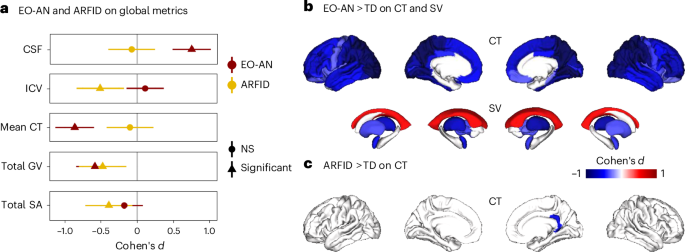
A widespread global pattern of thinner cortex (Cohen’s d = −0.86, Padj = 1.1 × 10−9), lower total gray matter volume (GV) (Cohen’s d = −0.58 Padj = 1.9 × 10−0.5) and cerebrospinal fluid (CSF) volume increase (Cohen’s d = 0.75, Padj = 5.9 × 10−8) was observed in patients with EO-AN compared with TD (Fig. 1a). We found no significant effect on the total surface area (SA) (Cohen’s d = −0.18, NS) or the intracranial volume (ICV) (Cohen’s d = 0.11, NS) in patients with EO-AN compared with TD. After accounting for BMI as a mediator, the EO-AN diagnosis was still associated with a lower CT (β = −0.781, P = 0.002) and a larger CSF volume (β = 0.668, P = 0.001), but no association remained significant with GV (Supplementary Table 37 and Supplementary Fig. 4). To assess the robustness of our findings, additional sensitivity analyses were conducted on subgroups of patients: (1) female individuals only (n = 106 EO-AN versus n = 57 TD), (2) participants scanned exclusively on a 3T MRI scanner (50 EO-AN versus 38 TD), (3) controls without a family history of psychiatric disorder (124 EO-AN versus 98 TD) and (4) participants without psychotropic medication (EO-AN (n = 101) versus TD (n = 116). The results remained consistent across all analyses (Supplementary Results and Supplementary Tables 26–31).

a, The effect of EO-AN (dark red dots, n = 124) and ARFID (yellow dots, n = 50) compared with TD (n = 116) on five global brain metrics (ICV, total SA, mean CT, CSF and total GV). The triangles represent significant effect sizes. Data are presented as mean values ±s.d. b,c, Brain maps (FDR thresholded) showing Cohen’s d values for each of the 68 cortical regions (thickness) and the 14 subcortical regions and ventricles (volumes) for EO-AN versus TD (b) and ARFID versus TD (c).
Regional effects
A widespread pattern of lower cortical thickness was identified, with a stronger contribution observed in the parietal and occipital lobes bilaterally. Fifty-nine regions showed significant thickness reduction (Fig. 1b and Supplementary Table 1). The regions with the higher effect sizes indicating thinner cortex included the left superior parietal cortex (Cohen’s d = −1.16, Padj = 8.9 × 10−15) and the precuneus (Cohen’s d = −1.01, Padj = 5.4 × 10−13) along with the right lingual (Cohen’s d = −1.01, Padj = 3.3 × 10−12). No significant regional alterations were found for SA (Supplementary Table 2). The results on CT and SA adjusted for global metrics are reported in the Supplementary Results (Supplementary Fig. 6a and Supplementary Tables 4 and 5). At the subcortical level, analyses comparing EO-AN and TD demonstrated smaller bilateral thalamic volumes (Cohen’s d = −0.71, Padj = 1.2 × 10−6 and Cohen’s d = −0.79, Padj = 1.1 × 10−7 for the left and right side, respectively), bilateral amygdala (Cohen’s d = −0.49, Padj = 7.7 × 10−4 and Cohen’s d = −0.41, Padj = 5.1 × 10−3 for the left and right side, respectively), bilateral pallidum (Cohen’s d = −0.38, Padj = 0.009 and Cohen’s d = −0.31, Padj = 0.03 for the left and right side, respectively) and bilateral putamen (Cohen’s d = −0.37, Padj = 0.009 and Cohen’s d = −0.36, Padj = 0.01 for the left and right side, respectively). We also observed larger bilateral ventricle volumes (Cohen’s d = 0.53, Padj = 3.3 × 10−4 and Cohen’s d = 0.50, Padj = 5.9 × 10−4 for the left and right side, respectively) (Fig. 1b and Supplementary Table 3). After accounting for BMI as a mediator, the EO-AN diagnosis remained significantly associated with lower CT in 32 regions, with the greatest effect observed in the superior parietal (Cohen’s d = −1.07, Padj < 0.0001). However, no subcortical volume (SV) differences remained significant (Supplementary Fig. 5 and Supplementary Tables 38 and 40).
We found an even stronger effect on CT (thinner) and SVs but still no effect on SA in the acutely ill subgroup of EO-AN patients (EO-acAN, BMI <3rd percentile), compared with TD (Fig. 2a and Supplementary Fig. 7).

a, The effects on CT and SV of EO-acAN > TD. b, The effects on SA and SV of unARFID > TD. c, The effects of the EO-acAN > unARFID on CT, SA and SV. d, Brain maps summarizing the 35 significant correlations between BMI and CT (68 cortical regions) as well as SV (14 subcortical regions + 2 lateral ventricles) in patients with EO-AN. No correlations were significant in patients with ARFID. e, Correlations between residuals (removing the effect of age, sex and scanner) of the thickness of the right middle temporal gyrus and z-scored BMI in individuals with EO-AN (dark red dots) and ARFID (yellow dots). We selected the right middle temporal gyrus as it showed the highest correlation with BMI in patients with EO-AN. Regression lines are shown with shaded gray areas representing 95% CIs. r represents Pearson correlation coefficient.
Brain pattern similarities between the EO versus TO types of AN
We found a correlation between EO-AN and TO-AN8 brain profiles at r = 0.78 (95% CI 0.67 to 0.86, P = 3.9 × 10−15) for the thickness of the 68 cortical regions and r = 0.88 (95% CI 0.68 to 0.95, P = 7.5 × 10−6) for the volume of the 16 subcortical regions. The main differences between EO-AN and TO-AN were observed in effect sizes for specific brain regions. For example, the transverse temporal gyrus showed a larger effect in EO-AN > TD (−0.87) compared with TO-AN > TD (−0.19), while the superior frontal gyrus had a greater effect in TO-AN > TD (−0.85) than EO-AN > TD (−0.63) (Supplementary Fig. 8). A comparison of the distributions of regional effect sizes showed a larger effect on CT in EO-AN > TD (absolute mean top decile of 1.06) compared with TO-AN > TD (absolute mean top decile of 0.89) and a larger effect on SVs in EO-AN > TD (absolute mean top decile of 0.79) compared with TO-AN > TD (absolute mean top decile of 0.66). Overall, these differences between EO-AN and TO-AN remain small and suggest a high degree of replication across independent cohorts, as well as signal stability on CT and SV metrics in AN, independent of the age of onset.
Distinct brain patterns associated with ARFID and EO-AN
ARFID was associated with smaller ICV (Cohen’s d = −0.51, Padj = 0.02), total GV (Cohen’s d = −0.48, Padj = 0.02) and total SA (Cohen’s d = −0.39, P = 0.04) compared with TD (Fig. 1a,c and Supplementary Table 12). At the regional level, only one region was significantly different between ARFID and TD: the thickness of the right isthmus of the cingulate gyrus (Cohen’s d = −0.62, Padj = 0.04). After including BMI as a mediator, significant differences between ARFID and TD were no longer observed (Supplementary Table 39). No effect on SA at the regional level was observed in ARFID > TD. When investigating specifically the underweight group of patients with ARFID (unARFID, BMI <3rd percentile), we observed a large reduction of the ICV (Cohen’s d = −0.68, Padj = 0.004), total GV (Cohen’s d = −0.69, Padj = 0.004) and total SA (Cohen’s d = −0.58, Padj = 0.01) (Supplementary Fig. 10). At the regional level, we observed lower SA in seven cortical regions (Fig. 2b and Supplementary Table 19). The top impacted regions were the right lingual gyrus (Cohen’s d = −0.72, Padj = 0.02), left postcentral area (Cohen’s d = −0.71, Padj = 0.02) and the right pericalcarine area (Cohen’s d = −0.71, Padj = 0.02). Finally, we observed a smaller left putamen volume (Cohen’s d = −0.65, Padj = 0.04) in unARFID > TD (Fig. 2b). Additional analyses on the ARFID clinical subtype ‘fear of aversive consequences’ (n = 29) were also performed, but no region survived false discovery rate (FDR) correction (Supplementary Fig. 9 and Supplementary Tables 33 and 34).
To disentangle the effects of the low BMI versus the eating disorders mechanisms, we compared patients with EO-acAN > patients with unARFID (both groups with a BMI percentile <3 at scan). We observed a similar profile to the one observed when comparing EO-acAN versus TD for thickness and volumes (r = 0.85, P < 2.2 × 10−16 and r = 0.82, P = 8.7 × 10−5, respectively; Fig. 2c), with a widespread lower CT and smaller SVs in EO-acAN compared with patients with unARFID. These results were similar when BMI was added as a covariate in the model (Supplementary Fig. 11). This suggests that the unARFID group resembles the TD group at the CT and SV levels. This was not the case for SA (r = 0.34, P = 0.004). We found a larger SA in three regions in patients with EO-acAN compared with patients with unARFID. These analyses demonstrate independent brain structural profiles associated with ARFID and EO-AN diagnoses despite similar BMI distributions.
To better understand the impact of weight recovery on brain features, we performed regional correlations between the residuals of each region of interest (n = 82 regions) and the z-scored BMI distribution at the individual level. The thickness of 35 cortical regions and the volume of three subcortical regions were positively correlated with BMI in subjects with EO-AN (Fig. 2d and Supplementary Table 35), showing that a z-scored BMI closest to 0 s.d. (closest to a normal weight) was associated with greater CT and volume. The highest correlation was observed for the middle temporal gyrus (r = 0.37, Padj = 0.001; Fig. 2e). In contrast, despite similar BMI distributions, we found no significant correlation for the CT or volume of any region in subjects with ARFID (Supplementary Table 35). No correlation between SA and BMI survived FDR correction in EO-AN or ARFID.
rEO-ED versus NDDs similarities at the brain and genetic levels
We extracted and ranked Cohen’s d distributions (Fig. 3a,b) for the brain signatures on CT of three neurodevelopmental conditions (OCD, ASD and ADHD) and compared them with EO-AN. EO-AN had the greatest effect on CT (mean absolute top decile of 1.06), followed by OCD (0.25), ASD (0.21) and ADHD (0.18). Brain-based Pearson correlations (rB) between EO-AN and these three conditions followed by permutation tests showed significant correlations between EO-AN and OCD (rB AN–OCD = 0.59, Pperm = 0.0001). Correlations between EO-AN and ADHD, as well as ASD, were not significant (rB AN–ADHD = −0.14 and rB AN-ASD = −0.03). We also performed similar correlation analyses between brain CT signatures of ARFID and these three conditions and found significant correlations between ARFID and ASD only (rB ARFID–ASD = 0.25, Pperm = 0.025 and rB ARFID–ADHD = −0.07, rB ARFID–OCD = 0.10). No significant correlations were found for SV patterns between EO-AN and ASD, OCD or ADHD. SA measures were not investigated as no significant results were found at the regional level in EO-AN > TD or ARFID > TD.

a, Distribution of effect sizes of three psychiatric conditions on regional CT (previously published by the ENIGMA consortium), as well as EO-AN (computed in this study). b, Brain maps representing patterns of abnormalities in CT reported by the ENIGMA consortium for three psychiatric conditions (Methods) and EO-AN. c, The relationship between single-nucleotide polymorphism (SNP)-based correlations (rG, provided by refs. 15,34) and brain-based correlations (rB, computed in this study using cortical thickness). Credit: brain and DNA icons in c, NIAID NIH BIOART.
Last, we observed a concordance of 0.94 between brain-based (CT) correlations of EO-AN and NDDs (computed in this paper) and genetic-based correlations of EO-AN and NDDs (previously published) across the three pairs of conditions (Fig. 3c).
link