Progression of gut microbiome in preterm infants during the first three months
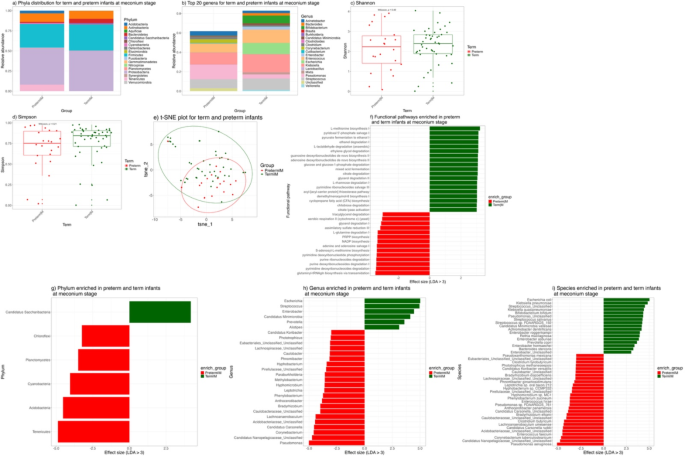
In our study, the most predominant phyla of meconium were Proteobacteria, Firmicutes, and Actinobacteria, in decreasing order of abundance, consistent with previous studies25. However, parallel studies reported differences in meconium’s prevalence of dominant phyla (e.g., Firmicutes, Proteobacteria, and Bacteroides)12,26,27,28. The variation is likely attributed to the geographic location, ethnicity, and gestational age. Analysis of the meconium microbiome revealed significant differences between term and preterm infants. Notably, in addition to the common phyla of Proteobacteria, Firmicutes, Bacteroides, and Actinobacteria, we found a relative predominance of Tenericutes and Acidobacteria in the meconium of preterm infants and a lower abundance of Bacteroidetes. Many species in the Bacteroidetes and Firmicutes exert nutritional or pharmacological effects by degrading polysaccharides in food, including Starch, glycogen, cellulose, pectin, etc., which are generally unable to be degraded by host digestive enzymes29, lower abundance of Bacteroidetes in preterm infants may be associated with impaired glucose metabolism in preterm infants30,31. Differences in the gut microbial composition at the phyla level may perform specific functions as part of the developing gut microecosystem, resulting in long-term impacts on the immune system or gut barrier function in preterm infants.
We found that the top 20 genera of preterm infants represented a lower proportion of overall diversity than term infants. This uneven structure of the meconium microbial composition of preterm infants may be related to the immature gut and immune function in preterm infants. The meconium of term infants was colonized by more bacteria from the digestive tract (e.g., Enterococcus, Escherichia, Bifidobacterium, Bacteroides, and Clostridiodes). In comparison, preterm infants were colonized by more bacteria from the skin and hospital environment (e.g., Corynebacterium, Cutibacterium, and Acinetobacter). Preterm infants in our study were predominantly delivered via cesarean section as well as were more exposed to maternal intrapartum antibiotics compared to term infants. Studies found that the gut microbiota of infants born by cesarean section is similar to that of maternal skin and hospital environment17, that a reduction in Bacteroides is a specific indicator of cesarean section delivery32, and that infants with intrapartum antibiotic exposure exhibit a diminished abundance of Bacteroides33. This might contribute to the differences in the meconium microbial composition between term and preterm infants in our cohort. Similar to the findings of Chernikova et al. (2018)15, a higher but statistically insignificant alpha diversity in meconium was observed in term vs. preterm infants. These findings are consistent with previous studies34, that preterm birth disrupted normal gut colonization, reducing diversity and altering the microbial composition.
As compared to preterm infants, the functional pathways of term infants were more enriched in L-methionine biosynthesis I, pyridoxal 5’-phosphate salvage I, pyruvate fermentation to ethanol I, and L-lactaldehyde degradation (anaerobic). While comparing with term infants at the meconium stage, the functional pathways of preterm infants were more enriched in glutaminyl-tRNAgln biosynthesis via transamidation, pyrimidine deoxyribonucleosides degradation, purine deoxyribonucleosides degradation I, and purine ribonucleosides degradation. Escherichia coli played an important role in amino acid metabolism35. Functional pathway indicated that term infants had increased L-methionine biosynthesis at birth, potentially associated with Escherichia (Escherichia coli). Some species of Streptococcus generated lactic acid during fermentation, which promoted gut health36. The increased prevalence of L-lactaldehyde degradation (anaerobic) in the functional pathway of term infants may be associated with Streptococcus (Streptococcus unclassified and Streptococcus salivarius). These functional pathways may correlate with a more stable microecological environment in term infants. While, the elevation of purine and/or pyrimidine deoxyribonucleoside or ribonucleoside in preterm infants may be related to elevated potential pathogen bacteria, like Pseudomonas (Pseudomonas aeruginosa) and Corynebacterium (Corynebacterium tuberculostearicum), some of which participate in cellular metabolic regulation37. The proliferation of pathogenic microbes and the decrease of beneficial microbes in preterm infants resulted in distinct functional pathways compared to term infants.
Furthermore, our study identified gestational age as the best explanatory variable contributing to differences in the initial microbial colonization profiles of meconium. This finding aligned with the positive correlation between gestational age and birth weight, highlighting the impact of early life events on gut microbiota development. Notably, the meconium microbiota in “extremely to very preterm” infants may contribute to delayed growth and development, and impaired intestinal immune and metabolic functions38. To the best of our knowledge, the study of microbiota in meconium is extremely rare in Malaysia. In this study, we did not detect significant differences in alpha and beta diversity reported elsewhere38,39. The lack of statistical significance might be attributed to the low sample size, but a trend of increasing alpha diversity with gestational age was observed.
Although most studies indicated that the gut microbiota of preterm infants predominantly consisted of Proteobacteria, Firmicutes, Actinobacteria, and Bacteroidetes, different patterns of microbial compositional development of preterm infants over time were observed. As gut microbes developed, Proteobacteria was gradually replaced by Actinobacteria and Bacteroidetes40. Bacteroidetes were consistently at low abundance13, while Actinobacteria were consistently at high prevalence in early life41. Previous studies have found that Bacteroides were enriched in most vaginally delivered infants but reduced in cesarean section infants. In our cohort, 82.6% of the preterm infants were delivered via cesarean section, and like the findings of Mitchell et al.42, we demonstrated that cesarean section preterm infants were born with the colonization of Bacteroides, but almost no Bacteroides was detected until the PNA of two months. The gut microbiota of preterm infants was strongly age-dependent. Some genera, such as Blautia, were virtually absent at early timepoints but became present in relatively high abundance from one month onwards. Over time, the microbiota progressed into a stable structure with Bifidobacterium as the predominant bacteria, while a reduction of Klebsiella was apparent.
Most studies have found that the diversity of gut microbes in preterm infants increased along with the PNA. Nevertheless, our study, like that of de Muinck and Trosvik43 found a non-linear progression of gut microbes in preterm infants, as well as a more homogeneous microbial composition with a higher abundance of Bifidobacterium breve, Bifidobacterium bifidum, and Bifidobacterium longum in our cohort from the PNA of one month to the PNA of three months. At the same time, this trend toward a more homogeneous microbial composition may be associated with increased breastfeeding rates in our cohort of preterm infants after discharge from the hospital between the PNA of one month and the PNA of three months. As with most studies, the specific developmental trajectories from the PNA of one month to the PNA of three months further confirmed a gradual increase in the colonization of preterm infants of the beneficial bacteria Bifidobacterium and Lactobacillus, and a gradual decrease of the potential pathogenic bacteria Clostridium, Enterobacter, Enterococcus, Klebsiella and Pseudomonas11.
Studies have shown that microbial diversity was lower in exclusively breastfed infants44,45. In contrast, formula-fed infants had a more diverse gut microbiota resembling a more mature gut microbiota45. This is consistent with our finding that breastfed preterm infants had the lowest α diversity compared to formula-fed and mixed-fed, allowing for a relatively higher abundance of beneficial bacteria. Our cohort of breast-fed infants had higher abundances of Bifidobacterium breve and Streptococcus sp. A12. Kordy et al.46 found Bifidobacteria breve in the rectum of a mother of a cesarean section infant, breast milk, and fecal from the infant. This finding suggested a potential in vivo hematogenous transfer pathway for entero-lactotransfer in breastfed infants, indicating that gut microbes can be established in infants by sharing the mother’s microbiome during breastfeeding. Ruiz-Ojeda et al.47 also found higher Bifidobacterium breve in breastfed, two-month-old infants. Fehr et al.48 demonstrated that Streptococcus spp. and Veillonella dispar co-existed in mother’s milk and infant feces and that the relative abundance of both genera was positively correlated between the abundance in breast milk and infant feces.
Antibiotics are essential medicines for perinatal healthcare and the treatment of newborns, especially preterm infants, and the survival rate during the perinatal period often depends on effective antibiotic treatment. Nevertheless, the use of antibiotics inevitably affects the host’s gut microbiota by reducing diversity49,50 and promoting the proliferation of antibiotic-resistance genes51. The mean total duration of antibiotic use for the preterm cohort throughout their NICU stay was 12.5 days (SD = 14.2). Reyman et al.50 revealed that different antibiotic regimens influenced gut microbes differently. However, the information on the antibiotic used was non-retrievable, so the association of microbial composition with the specific antibiotic types was impossible. As per the findings of Zwittink et al.52, we found that the duration of antibiotic treatment was the primary determinant of gut microbial composition, with infants showing a high degree of variability between and within infants across different durations of antibiotic treatment. In the study by Zwittink et al.52, infants who received prolonged antibiotic treatment were mainly manifested by a decrease in Bifidobacterium and an increase in Enterococcus.
We detected a lower microbial diversity in preterm infants diagnosed with diseases (IVH, BPD, NEC, RDS, ROP, PDA, Nosocomial pneumonia). However, only differences in PDA and gut microbiota were significantly associated in our cohort, although associations between gut microbiota and NEC and sepsis have been reported in other cohorts21,53. Our cohort of PDA infants had decreased α diversity of gut microbiota, while Streptococcus oralis and Streptococcus mitis were the domain species that were differentially abundant in preterm infants with PDA. Among the five preterm infants with PDA, the duct sizes ranged from 0.8 mm to 3.7 mm, and three of them had shunting across the duct, leading to hemodynamic disturbance, i.e., hemodynamically significant PDA (hsPDA). Ventricular septal defect, atrial septal defect, and PDA are the three most common types of congenital heart disease. Gut microbiota abnormalities may be associated with hemodynamic abnormalities in preterm infants with hsPDA. Infants with hsPDA have an aortopulmonary shunt and may have systemic hypoperfusion, which causes altered intestinal hemodynamics and disturbances in peritoneal or mesenteric blood flow54. To our knowledge, there are no studies on the gut microbiota of infants with PDA. However, studies by Zhang et al.55 and Huang et al.56 found that the composition of the gut microbiota was significantly disturbed in infants with critical congenital heart disease compared to non-cardiac infants. In our study, the prevalence of Streptococcus oralis and Streptococcus mitis was significantly elevated in infants with PDA; this may indicate a potential association between specific species and clinical outcomes. This finding highlights a potential link between cardiovascular health and gut microbiota in preterm infants, and there is a need to further investigate the underlying mechanisms of this link.
This study has several limitations. First, the lack of detailed information on the specific types of antibiotics administered to preterm infants constrained our ability to analyze the association between gut microbiota and particular antibiotic regimens. Second, because of the relatively small sample size, n = 23 (five late preterm, nine moderate preterm, six very preterm, and three extremely preterm), we reclassified the samples into “extremely to very preterm,” “moderate to late preterm,” which limited our capacity to elucidate the relationship between variations in more detailed gestational age categories and differences in the meconium microbiota. At the same time, the meconium samples in our study were collected either as first-pass meconium or within 72 h postnatal. Nevertheless, our results on the composition of meconium microbiota were consistent with previously published studies of larger cohorts25. Therefore, we believe that these samples are representative of the gut microbial composition of the studied infants. Finally, while we identified a significant association between PDA and gut microbiota within a longitudinally sampled cohort, the generalizability of these findings requires validation in larger, more diverse cohorts. Expanding the sample size and breadth of future studies will enhance statistical power and facilitate a better understanding of the associations between individual developmental trajectories and relevant health outcomes.
The most predominant phyla in meconium were Proteobacteria, Firmicutes, and Actinobacteria, which were thought to be the early gut colonizers of infants, possibly reflecting microbial exposure in utero57. Compared to preterm infants, the meconium microbiota of term infants showed higher diversity and differentially enriched taxa. The gut microbiota of preterm infants predominantly consisted of Proteobacteria, Firmicutes, Actinobacteria, and Bacteroidetes. As gut microbes developed, Proteobacteria was gradually replaced by Actinobacteria and Bacteroidetes. The gut microbiota of preterm infants was strongly age-dependent and progressively developed into a more homogeneous gut microbial structure with Bifidobacterium as the predominant bacteria. At the same time, a decrease in Klebsiella was apparent. Divergences in gut microbiota may significantly influence functional pathways at birth. The taxonomic composition of the meconium microbiota of infants was primarily associated with gestational age. Additionally, variations in the gut microbiota of preterm infants at the PNA of three months were linked to individualized treatments, including feeding methods and the duration of antibiotic therapy. Also, we found lower microbial diversity in preterm infants diagnosed with preterm birth-related complications, especially the association between PDA and gut microbiota. Preterm infants with PDA had a higher abundance of Streptococcus oralis and Streptococcus mitis, which, to the best of our knowledge, has not been reported before, highlighting the role of gut microbiota in disease. Our study further refines the association between PDA, an early-life congenital heart disease, and the gut microbiota as a clinical non-invasive diagnostic biomarker of congenital heart disease, providing a basis for developing non-invasive diagnostic tests and personalized intervention strategies, thereby improving clinical outcomes.
link